Frequently Asked Questions
On our FAQ page, here you will find answers to the most common questions about using SkinScreener and skin health in general. We know there can be a lot of questions when it comes to tracking changes on your skin. That’s why we want to make sure you have all the information you need to use our app successfully.
About SkinScreener
What is the motivation behind SkinScreener?
We monitored that the incidence of skin cancer has increased steadily worldwide in recent years. Statistically, the probability of developing skin cancer once in a lifetime is about 20%. The earlier skin cancer can be detected, the better are the chances of recovery. This is why we, with all our expertise from medical and technical backgrounds, have developed SkinScreener. We wanted to offer a smart way to determine the personal risk of altered skin areas with an App.
I'm a journalist. Where can I get press information?
We are more than happy to answer your questions and send you information. Do not hesitate to send us your request for an interview. You can contact us via the contact page.
Where is SkinScreener available?
At the moment the app is available in Germany, Austria, Great Britain and Ireland.
General
SkinScreener is a Class I medical device. What does that mean?
According to EU Regulation 2017/745 (MDR) Chapter I, Article 2/1, a medical device is an instrument, apparatus, device, software, implant, reagent, material, or other article that, according to the manufacturer, is intended for human use and is intended, alone or in combination, to fulfil one or more of the following specific medical purposes:
Diagnosis, prevention, monitoring, prediction, prognosis, treatment or alleviation of disease,
Diagnose, monitor, treat, mitigate, or compensate for injury or disability,
Examination, replacement, or alteration of anatomy or of a physiological or pathological process or condition,
Obtaining information through the in vitro study of from the human body.
including samples from organ, blood and tissue donations
and whose principal intended action in or on the human body is not achieved by pharmacological or immunological means or metabolically, but whose mode of action may be assisted by such means.
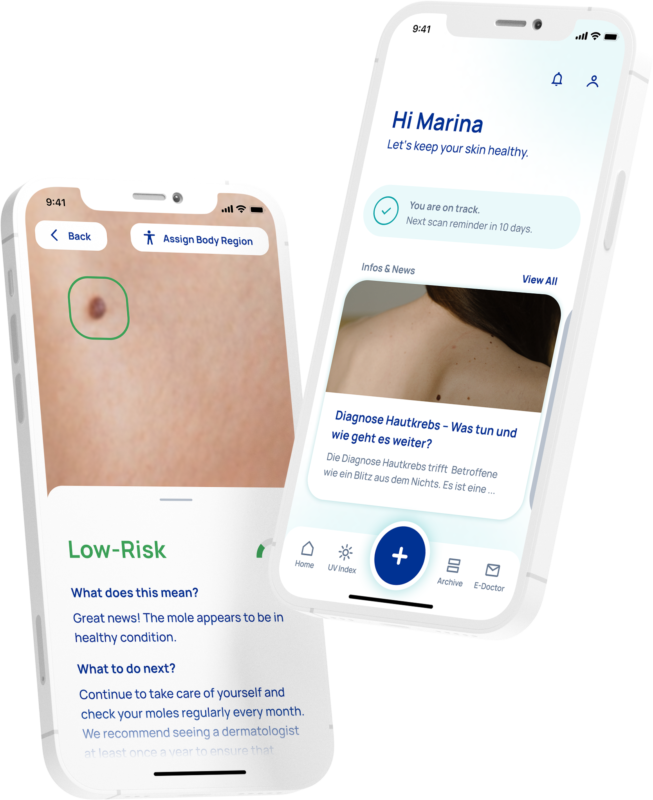
How does the App work?
In the App, you are asked to take a photo of the designated skin area with the camera of your smartphone. The image is then run through our clinically proven neural network. You immediately receive the result. The risk assessment for skin cancer is indicated by an easy to understand 3 color code (low, medium, high risk). Green means low risk, yellow means medium risk, and red means high risk.
What is a skin lesion?
A skin lesion is a part of the skin that has an abnormal growth or appearance compared to the skin around it. In medicine, the term lesion usually refers to all types of skin changes and skin damage.
Why is early detection of skin cancer important?
One in five people develop skin cancer. Skin cancer and its preliminary stages are visible and palpable. Therefore, regular self-examinations can help you discover early on where new skin changes have appeared or existing moles have grown. The earlier skin cancer is detected, the higher the chances of cure.
Can anyone use SkinScreener?
Almost anyone can use SkinScreener. The app is intended for adult users with Fitzpatrick skin type I (ivory) to type IV (tan). SkinScreener should not be used on children.
Can I use SkinScreener on any smartphone?
Yes, however, the technical requirements for using SkinScreener are iOS operating system version 13.0 or later or Android operating system version 9.0 or later. In order to have a correct risk assessment, a good working smartphone camera and flash is required. To ensure that the camera quality is good enough, a camera test must be performed before you can start scanning.
Does SkinScreener replace the doctor?
The app serves as a precautionary instrument and as such offers numerous added values for people. The accuracy rate is over 95%. However, the medium as well as high skin lesions should be examined by a specialized dermatologist.
Who can I contact with questions?
For questions, email support@skinscreener.at
Further app functions
How does the UV index work?
The UV index displays the current weather situation as a numerical value.
How does the image archive work?
Medium and high risk skin lesions are stored in the archive. Double click on the image to see the lesion in full size.
My account
Is it safe to use SkinScreener?
Yes. SkinScreener was developed and clinically tested by a team of dermatologists, physicians and biomedical engineers. SkinScreener is a Class I medical device and complies with the requirements of the European Directive 93/42/EEC. A clinical study at the Medical University of Graz (using more than 600 images of real patients) has proven that SkinScreener has an impressive accuracy rate of 95% in the detection of skin cancer. We recommend reading the Instructions for Use in its entirety prior to first use.
Is my personal information safe?
Yes, the protection of personal data with respect to data processing is important to medaia GmbH as the data controller. Consequently, when using personal data, medaia GmbH complies with all the provisions of the General Data Protection Regulation (GDPR) and the Data Protection Act (DSG) and strives for the best possible transparency. medaia GmbH processes personal data in accordance with the principles of lawfulness, good faith, transparency, accuracy, purpose limitation, data minimization, and storage limitation.
Does my purchased subscription automatically renew after the duration of the subscription is over?
The duration of the contract as well as the notice periods and deadlines for the subscription package are based on the terms of use of the Google or Apple App Stores.
I can't download SkinScreener from my App Store.
Reasons for this can be that your Smartphone does not fulfill the system requirements, there is not enough processing power or storage available. SkinScreener is currently available in Austria, Germany, the United Kingdom and Ireland.
How can I sign in to the App with my Apple/Google account?
After accepting the Privacy Policy and the Terms and Conditions, you should click on the “Log in with Apple/Google” button to successfully complete the registration process.
Why do I need to sign in with my Apple ID?
Signing in with your Apple ID is the fastest, easiest, and most confidential way to sign in to third-party apps and websites with an existing Apple ID. Apple Login protects your privacy and is designed to help you maintain control over your personal information. You can find more information here: https://support.apple.com/en-gb/HT210318
Why do I need to sign in with my Google Account?
To prevent unauthorized access, Advanced Protection only allows Google apps and verified third-party apps to access your Google Account data, and only with your permission. Google Account Login protects your privacy and is designed to help you stay in control of your personal information. You can find more information here: https://support.google.com/accounts/answer/112802?hl=en&co=GENIE.Platform%3DAndroid&oco=1
Pricing
Why do I have to pay for the service?
SkinScreener is a Class I medical device and complies with the requirements of the European Directive 93/42/EEC. In order to ensure the highest possible technical and medical safety, our team of doctors and technicians is constantly working to ensure and further develop our quality management system, which is associated with high costs. By using SkinScreener, you can initiate medical treatment at an early stage through the appropriate risk assessment and reduce the progression of skin cancer and its preliminary stages. We offer the first two scans for free, so you can download the App and see how it works without paying for it. If you would like to do more scans, you can pick a subscription model which fits you best.
How much does SkinScreener cost?
SkinScreener can be downloaded for free from the App Store and after registering you get two free scans. After that, you can choose from three different plans. To do this, visit www.skinscreener.com/pricing
The risk assessment
How does risk assessment work?
SkinScreener’s risk assessment is based on a clinically validated algorithm developed and tested in collaboration with the Medical University of Graz. The algorithm works with a neural network that uses artificial intelligence to ensure a risk assessment with 95% accuracy.
What does it mean to have low, medium or high risk?
If your scan result shows a LOW RISK, we advise to monitor the spot on your skin regularly and rescan it at least once a month. If your scan result shows a MEDIUM RISK or HIGH RISK rating, it means you should see your dermatologist as soon as possible and earlier than the recommended dermatological examinations. The specialist will then verify the results by further specialist examinations.
What if I get a high-risk assessment?
If your scan result shows a HIGH RISK rating, it means you should see your dermatologist as soon as possible. The specialist will then verify the results by further specialist examinations.
Why is it important to scan monthly?
As skin spots evolve over time, it is important to keep track of these spots. Look out by using SkinScreener as gradual changes in shape, size or color may lead to a new risk assessment.
It seems there is no risk assessment of my scanned skin lesion possible. Why did the scanning not work?
Usually, most inadequate risk assessments are due to the incorrectly scanned skin lesions. Lesions next to anatomical structures are sometimes hard to detect. Make sure the lesion is focused and the distance between the camera and the skin area is approximately 15 cm. Zoom in or zoom out a skin lesion, tap and hold the screen to scan the skin area. Additionally, make sure that nothing is on blocking the view on your designated skin area. Try to find an angle to avoid having a nose, eye, hair, mouth, nipples, fingers or toes, blocking the view on the suspected skin area.
Easy to use.
Everywhere and at any time.
Have the cancer risk of moles assessed quickly, easily and accurately anywhere, anytime with SkinScreener.
*Apple App Store available in a few days!
- SkinScreener cannot replace a dermatological examination. SkinScreener does not diagnose and is not a substitute for regular visits to dermatologists.